This fragmentation means that data extraction, aggregation, and analysis still rely heavily on manual processes, requiring substantial time and human resources. As the health sector is increasingly pursuing digital transformation, the development of an integrated data model capable of connecting information across hospital information systems (HIS) has become increasingly necessary. Such an approach would allow better use of existing systems while supporting more timely, data-driven management.
Building on these practical needs, within the framework of the 60HN–ASPARNet study, OUCRU developed iSHARP (Integrated Stewardship in Hospitals for Antimicrobial Resistance Control Programme), an integrated data platform designed to support data-driven AMS and IPC in hospitals. iSHARP is designed as a solution that helps hospitals consolidate data into a unified reporting system, providing hospital leadership with timely access to data‑driven insights to support informed decision‑making. iSHARP does not replace existing HIS. Instead, it serves as a layer that links and brings together data from current systems while preserving routine data entry workflows for healthcare staff. The platform can connect prescribing data from electronic medical record systems, connect laboratory results from laboratory information systems, and incorporate IPC data through electronic forms where available. With this approach, iSHARP can be implemented flexibly, minimizing changes to established work practices and avoiding disruption to routine hospital operations.
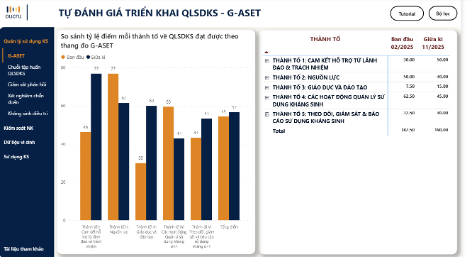
On this integrated data platform, iSHARP is built around multiple functional components that together provide comprehensive support for AMR control and patient safety initiatives. First, the platform helps hospitals review and assess ongoing AMS activities, such as training, audit and feedback, allowing them to identify what has been implemented and where further improvement is needed. At the same time, iSHARP supports the management and monitoring of IPC activities by systematically compiling relevant information and indicators. The platform also integrates microbiology data management, enabling the development and monitoring of cumulative antibiogram reports through tables and visual displays that reflect trends in AMR over time. In addition, iSHARP supports AMS based on prescribing data from electronic medical record systems, allowing hospitals to track antimicrobial use indicators by department and over different periods, and to establish a unified data foundation for both clinical practice and management purposes.
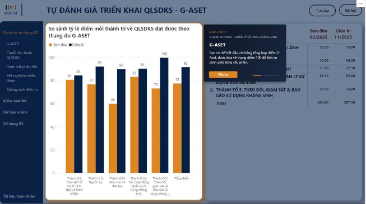
At the hospital level, iSHARP helps significantly reduce the workload associated with data aggregation and reporting. Instead of extracting data from multiple systems and processing them separately through manual means, the platform supports the automated consolidation and visualization of information through tables and charts.
iSHARP provides hospital leadership with a clear, comprehensive, and timely overview of AMS and IPC activities, supporting data‑informed leadership, monitoring, and decision‑making. For AMS teams, the platform enables monitoring of antimicrobial use trends by department, assessment of adherence to treatment recommendations, and early identification of issues requiring intervention. Meanwhile, IPC teams can use systematically aggregated and clearly presented data to monitor trends in hospital‑acquired infections and to support the implementation and adjustment of appropriate control measures.
More importantly, iSHARP provides a unified data foundation to support clinical decision-making, allowing clinicians to refer to antimicrobial use patterns and resistance trends by department during the treatment process. This information contributes to improving the appropriateness of antimicrobial use, reducing the risk of hospital-acquired infections, and enhancing patient safety.
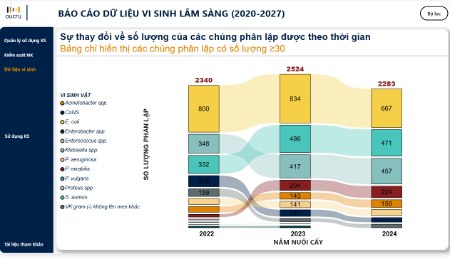
At the management level, iSHARP offers Departments of Health (DoH) the opportunities to gradually strengthen more proactive oversight and coordination, rather than relying solely on periodic reports submitted by hospitals. The platform allows real-time monitoring of antimicrobial use, resistance trends, and IPC activities across multiple healthcare facilities. This supports early identification of facilities that may require prioritized support or intervention. Based on systematically aggregated and clearly presented data, iSHARP has the potential to support DoH in developing training plans, allocating resource, and organizing supervisory activities aligned with actual needs. At the same time, the platform contributes to standardizing indicators related to AMS and IPC at the provincial level, creating a data foundation for reporting, supporting management functions, and facilitating the implementation of AMR prevention and control activities.
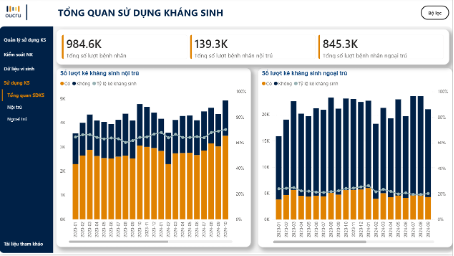
Beyond its value for hospitals and health authorities, iSHARP also opens up opportunities for collaboration with HIS providers. By adding an advanced data analysis layer and in-depth visual reporting, iSHARP enhances the usability and added value of existing systems, while also expanding the potential to develop specialized solutions for AMS and IPC. Currently, a comprehensive iSHARP prototype is being piloted at two hospitals and with the plan for gradual expansion to other healthcare facilities in the region under the direction of DoH. This approach aligns with the development of sustainable digital health solutions, allowing for testing, refinement, and step-by-step scale-up in real-world settings.With a design philosophy focused on avoiding system disruption, maximizing the use of existing data, and visualizing key indicators in line with international recommendations, iSHARP represents an important step toward data-driven management for hospitals and Departments of Health. Developed by OUCRU within the framework of the 60HN–ASPARNet study, iSHARP is not merely a technical tool but a platform that supports meaningful digital transformation. It contributes to improving the quality of patient care, strengthening infection control, and promoting more rational use of antimicrobials in Viet Nam.
















